Abstract
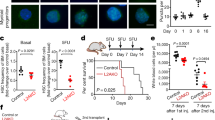
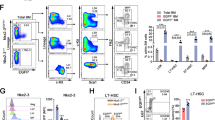
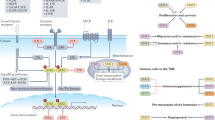
Haematopoietic stem cells (HSCs) can convert between growth states that have marked differences in bioenergetic needs. Although often quiescent in adults, these cells become proliferative upon physiological demand. Balancing HSC energetics in response to nutrient availability and growth state is poorly understood, yet essential for the dynamism of the haematopoietic system. Here we show that the Lkb1 tumour suppressor is critical for the maintenance of energy homeostasis in haematopoietic cells. Lkb1 inactivation in adult mice causes loss of HSC quiescence followed by rapid depletion of all haematopoietic subpopulations. Lkb1-deficient bone marrow cells exhibit mitochondrial defects, alterations in lipid and nucleotide metabolism, and depletion of cellular ATP. The haematopoietic effects are largely independent of Lkb1 regulation of AMP-activated protein kinase (AMPK) and mammalian target of rapamycin (mTOR) signalling. Instead, these data define a central role for Lkb1 in restricting HSC entry into cell cycle and in broadly maintaining energy homeostasis in haematopoietic cells through a novel metabolic checkpoint.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Change history
06 July 2011
Figs 1 and 2 have been corrected in the original online HTML and PDF versions as described in the accompanying Erratum.
References
Tothova, Z. & Gilliland, D. G. FoxO transcription factors and stem cell homeostasis: insights from the hematopoietic system. Cell Stem Cell 1, 140–152 (2007)
Gan, B. et al. mTORC1-dependent and -independent regulation of stem cell renewal, differentiation, and mobilization. Proc. Natl Acad. Sci. USA 105, 19384–19389 (2008)
Ito, K. et al. Regulation of reactive oxygen species by Atm is essential for proper response to DNA double-strand breaks in lymphocytes. J. Immunol. 178, 103–110 (2007)
Liu, J. et al. Bmi1 regulates mitochondrial function and the DNA damage response pathway. Nature 459, 387–392 (2009)
Chen, C. et al. TSC-mTOR maintains quiescence and function of hematopoietic stem cells by repressing mitochondrial biogenesis and reactive oxygen species. J. Exp. Med. 205, 2397–2408 (2008)
Shackelford, D. B. & Shaw, R. J. The LKB1-AMPK pathway: metabolism and growth control in tumour suppression. Nature Rev. Cancer 9, 563–575 (2009)
Shaw, R. J. et al. The tumor suppressor LKB1 kinase directly activates AMP-activated kinase and regulates apoptosis in response to energy stress. Proc. Natl Acad. Sci. USA 101, 3329–3335 (2004)
Hawley, S. A. et al. Complexes between the LKB1 tumor suppressor, STRADα/β and MO25α/β are upstream kinases in the AMP-activated protein kinase cascade. J. Biol. 2, 28 (2003)
Hardie, D. G. AMP-activated/SNF1 protein kinases: conserved guardians of cellular energy. Nature Rev. Mol. Cell Biol. 8, 774–785 (2007)
Narbonne, P. & Roy, R. Caenorhabditis elegans dauers need LKB1/AMPK to ration lipid reserves and ensure long-term survival. Nature 457, 210–214 (2009)
Lizcano, J. M. et al. LKB1 is a master kinase that activates 13 kinases of the AMPK subfamily, including MARK//PAR-1. EMBO J. 23, 833–843 (2004)
Kiel, M. J. et al. SLAM family receptors distinguish hematopoietic stem and progenitor cells and reveal endothelial niches for stem cells. Cell 121, 1109–1121 (2005)
Bardeesy, N. et al. Loss of the Lkb1 tumour suppressor provokes intestinal polyposis but resistance to transformation. Nature 419, 162–167 (2002)
Kuhn, R., Schwenk, F., Aguet, M. & Rajewsky, K. Inducible gene targeting in mice. Science 269, 1427–1429 (1995)
Chan, I. T. et al. Conditional expression of oncogenic K-ras from its endogenous promoter induces a myeloproliferative disease. J. Clin. Invest. 113, 528–538 (2004)
Torchia, E. C., Boyd, K., Rehg, J. E., Qu, C. & Baker, S. J. EWS/FLI-1 induces rapid onset of myeloid/erythroid leukemia in mice. Mol. Cell. Biol. 27, 7918–7934 (2007)
Essers, M. A. G. et al. IFNα activates dormant haematopoietic stem cells in vivo . Nature 458, 904–908 (2009)
Maiuri, M. C., Zalckvar, E., Kimchi, A. & Kroemer, G. Self-eating and self-killing: crosstalk between autophagy and apoptosis. Nature Rev. Mol. Cell Biol. 8, 741–752 (2007)
Ichimura, Y. et al. A ubiquitin-like system mediates protein lipidation. Nature 408, 488–492 (2000)
Shaw, R. J. et al. The LKB1 tumor suppressor negatively regulates mTOR signaling. Cancer Cell 6, 91–99 (2004)
Corradetti, M. N., Inoki, K., Bardeesy, N., DePinho, R. A. & Guan, K. L. Regulation of the TSC pathway by LKB1: evidence of a molecular link between tuberous sclerosis complex and Peutz-Jeghers syndrome. Genes Dev. 18, 1533–1538 (2004)
Shaw, R. J. et al. The kinase LKB1 mediates glucose homeostasis in liver and therapeutic effects of metformin. Science 310, 1642–1646 (2005)
Koh, H. J. et al. Skeletal muscle-selective knockout of LKB1 increases insulin sensitivity, improves glucose homeostasis, and decreases TRB3. Mol. Cell. Biol. 26, 8217–8227 (2006)
Koh, H. J. et al. Sucrose nonfermenting AMPK-related kinase (SNARK) mediates contraction-stimulated glucose transport in mouse skeletal muscle. Proc. Natl Acad. Sci. USA 10.1073/pnas.1008131107 (16 August 2010)
Gurumurthy, S. et al. LKB1 deficiency sensitizes mice to carcinogen-induced tumorigenesis. Cancer Res. 68, 55–63 (2008)
Contreras, C. M. et al. Loss of Lkb1 provokes highly invasive endometrial adenocarcinomas. Cancer Res. 68, 759–766 (2008)
Hezel, A. F. et al. Pancreatic LKB1 deletion leads to acinar polarity defects and cystic neoplasms. Mol. Cell. Biol. 28, 2414–2425 (2008)
Sanchez-Cespedes, M. A role for LKB1 gene in human cancer beyond the Peutz-Jeghers syndrome. Oncogene 26, 7825–7832 (2007)
Klimova, T. A. et al. Hyperoxia-induced premature senescence requires p53 and pRb, but not mitochondrial matrix ROS. FASEB J. 23, 783–794 (2009)
Ozsolak, F. et al. Direct RNA sequencing. Nature 461, 814–818 (2009)
Hezel, A. F. & Bardeesy, N. LKB1; linking cell structure and tumor suppression. Oncogene 27, 6908–6919 (2008)
Nakada, D., Saunders, T. L. & Morrison, S. J. Lkb1 regulates cell cycle and energy metabolism in haematopoietic stem cells. Nature 10.1038/nature09571 (this issue)
Gan, B. et al. Lkb1 regulates quiescence and metabolic homeostasis of haematopoietic stem cells. Nature 10.1038/nature09595 (this issue)
Watson, M. et al. The small molecule GMX1778 is a potent inhibitor of NAD+ biosynthesis: strategy for enhanced therapy in nicotinic acid phosphoribosyltransferase 1-deficient tumors. Mol. Cell. Biol. 29, 5872–5888 (2009)
Ventura, A. et al. Restoration of p53 function leads to tumour regression in vivo . Nature 445, 661–665 (2007)
Ferrick, D. A., Neilson, A. & Beeson, C. Advances in measuring cellular bioenergetics using extracellular flux. Drug Discov. Today 13, 268–274 (2008)
Robinson, M. D. & Oshlack, A. A scaling normalization method for differential expression analysis of RNA-seq data. Genome Biol. 11, R25 (2010)
Acknowledgements
We would like to thank the Harvard Stem Cell Institute Flow Cytometry Core and D. Brown and the MGH Electron Microscope Imaging Core for expertise and advice. V. Levy and M. Leisa provided technical assistance in the SeaHorse measurements. D. Van Buren provided advice and assistance in pilot experiments. We thank A. Camacho for mouse colony assistance. We would also like to thank A. Kimmelman, R. Mostoslavsky, M. Vander Heiden, L. Ellisen, W. Kim and M. Ivan for discussions and critical review of the manuscript. We are grateful to S. Morrison and R. DePinho for sharing unpublished data. N.B. would like to acknowledge support from NIH U01 CA141576-01. D.T.S would like to acknowledge support from NIH DK050234 and Ellison Medical Foundation. S.G. was supported by a Massachusetts Biotechnology Research Council Tosteson Fellowship. S.Z.X. was supported by an NIH Ruth L. Kirschstein National Research Service Award.
Author information
Authors and Affiliations
Contributions
S.G. and S.Z.X. performed the experiments with assistance from B.A., J.K., A.T., B.S. and R.Z.Y. S.G., S.Z.X., D.T.S. and N.B. designed the experiments, analysed and evaluated all data, and wrote the manuscript. F.O. and P.M. performed the RNA sequencing. F.F. and P.J.P. analysed the RNA sequencing data. O.S.S. designed and evaluated the oxygen consumption experiments.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Figures
This file contains Supplementary Figures 1-11 with legends. (PDF 745 kb)
Rights and permissions
About this article
Cite this article
Gurumurthy, S., Xie, S., Alagesan, B. et al. The Lkb1 metabolic sensor maintains haematopoietic stem cell survival. Nature 468, 659–663 (2010). https://doi.org/10.1038/nature09572
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature09572
This article is cited by
-
Induction of mitochondrial recycling reverts age-associated decline of the hematopoietic and immune systems
Nature Aging (2023)
-
Effects of smoking on the tissue regeneration-associated functions of human endometrial stem cells via a novel target gene SERPINB2
Stem Cell Research & Therapy (2022)
-
Myeloid liver kinase B1 contributes to lung inflammation induced by lipoteichoic acid but not by viable Streptococcus pneumoniae
Respiratory Research (2022)
-
Endothelial cell-specific expression of serine/threonine kinase 11 modulates dendritic cell differentiation
Nature Communications (2022)
-
Novel roles of luteinizing hormone (LH) in tissue regeneration-associated functions in endometrial stem cells
Cell Death & Disease (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.